This Class for
- Physician
- Resident / Fellow
What I will learn?
Enhance awareness and clinical understanding among targeted healthcare professionals regarding Atopic Dermatitis (AD) and the differentiated role of Ebglyss (lebrikizumab) in the management of moderate-to-severe AD.
Disclaimer
This content is approved by Eli Lilly and Company (“Lilly”). This content is intellectual property of Lilly and its reproduction needs Lilly's prior permission
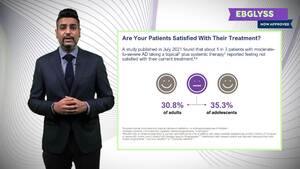
(This information/content is intended to Health Care Providers only)
For adverse events and safety reporting: email: [email protected]
24 /7 mobile: +966 567 868 843, Working hours Tel: +966 11 461 7800 , Ext: 1
For Lilly product information: email: [email protected]
For further information, please contact:
Eli Lilly and Company, Scientific Office P.O. Box 92120 Riyadh 11653, Kingdom of Saudi Arabia. Tel: +966 11 461 7800 Fax: +966 111 217 9900 email: [email protected]
Please go to PDF Material for prescribing information.
PP-LK-SA-0064